

Think of a burning match, or a fire in the fireplace or a roaring campfire. In each case you have felt the heat given off as these materials burn (but hopefully not as a close encounter!). Where does this heat (energy) come from?
The reactants, such as the wood in the campfire, have a certain amount of energy stored in their chemical bonds. This energy can be released when these materials react with the oxygen in the air (combustion) and are converted to the combustion products (usually carbon dioxide and water). Chemists use a quantity called the heat of formation as a measure of the energy stored in these compounds. The energy change associated with a combustion reaction is the difference between the energy (heat of formation) of the products and the energy (heat of formation) of the reactants.
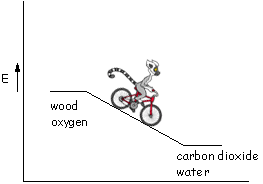
In an combustion reaction, the products are at a lower energy than the reactants. During the combustion process we go from a higher energy state to a lower energy state, just like riding a bike downhill. In the chemical process, going "downhill" means releasing energy, usually in the form of heat. (Reactions that release heat are exothermic.) The larger the "hill" the more energy that is released (remember this - it's important later!).
The field of thermochemistry involves the study of the energy changes associated with chemical process (such as combustion). The "heat of combustion" (or enthalpy of combustion) is the amount of heat released by a combustion reaction. That is, it's the amount that we travel "down the energy hill" as the material burns.
So, if the thermochemistry says wood is in a much higher energy state than its combustion products, why doesn't it spontaneously ignite? Think about it.
So, the science of burning materials is pretty simple isn't it? Well, not really. In addition to the heat that's released, there are many factors to consider, such as kinetics. Kinetics is the measure of how fast a reaction takes place (or how fast the wood burns). The change in the energy diagram above is sort of like coasting on a bike - it's all downhill. As we all know, before you can coast downhill you need to go uphill. In kinetics the "uphill" part is the activation energy.
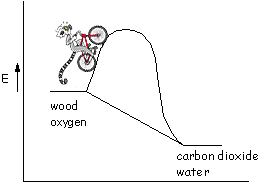
What determines the activation energy in the combustion process? In a combustion reaction, the activation energy is the energy needed to get the small molecules into the gas phase. Obviously, if there is not enough energy to get these molecules, the fuel, into the air then the fire will not start. The stronger the bonds in the material, the harder it is to get the fuel into the gas phase and the larger the activation energy. When we use a heat source to ignite a fire, we're suppling the energy needed to overcome the activation energy to start the combustion process. Once the process starts, there needs to be a continuous source of energy to let us keep "climbing the activation energy hill".
Food accidently heated very hot in a covered pan seldom bursts into flame, but food catches fire all too easily in an open frying pan. What is going on?
Fuel:
The combustion process requires a fuel. So what's the fuel? In burning wood, the fuel really isn't the wood, but rather small molecules that are released from the wood's surface, as gases, when the wood is heated. It's these gases that react with oxygen in the air to produce the combustion products and heat.
The reason that heat is required to start a fire is because we need to supply enough energy to get these small molecules into the air around the wood. Once these molecules are in the air, they need to be ignited. A flame or a spark does the trick if the gas temperature is above the flash-ignition temperature. Materials will still burst into flames if you get them hot enough. Just get the temperature above the self-ignition temperature. Where do you think the energy to keep the fuel burning comes from?. I'm sure that you can figure it out now. (But we'll still tell you a lot more latter.)
In order for rapid combustion (flames!) to take place, both the fuel and the oxidant (usually the oxidant is gaseous oxygen from the atmosphere) must be thoroughly mixed together. If the frying pan is heated while covered, the gases force the air (oxygen) out of the pan as they are produced. Without an oxidant - no flames!
Unfortunately, if the lid is removed while the pan is hot, the contents may suddenly flash and catch fire. This rapid combustion produces a great deal of heat (energy) that keeps the cycle going. That's why it's always best to remove a covered pan from the heat for a few minutes before it's uncovered. That way, the hot gases are less likely to catch fire or cause burns.
What do these observations suggest for controlling or halting a fire? Think about the stages of fire you've just learned.
Now for the fun part. Let's think about combustion:

|

|