
Keywords
covalent bond,
secondary interaction,
hydrogen bond,
strength
Have you ever realized how many times the word gel is heard in your everyday life?
If you work in a lab, probably you're familiarized with such terms like silica gel, gel electrophoresis, or gel permeation chromatography.
Otherwise, you can be absolutely sure that such "gels" will have nonetheless entered your life without any notice. Just take a look at what the market does offer these days:

One can assume that gels' application range is as huge as the inventors' imagination! And that's right: gels are everywhere.
But if you have a curious, inquisitive mind, it is possible you may want to know what gels exactly are. I mean they are polymers, obviously, but what kind of polymers? How can they be obtained? Why are they so intensely commercialized in the market?
The first thing you must bear in mind when speaking about gels is crosslinking. As you already know, crosslinked polymers are insoluble in all solvents, but they can be swollen at different extent, depending on their crosslinking degree. But how polymers become crosslinked within a solution?
Concentrated Solutions
Remember what we stated previously about polymer solutions? Depending on the amount of dissolved polymer, solutions can be dilute or concentrated. Take a look at these pictures:
 |
 |
|---|
There is no interaction at all between the dissolved coils in dilute solutions, as shown in the figure on the left. Even when the solution is highly viscous, such coils remain as unique entities moving freely among the solvent molecules, without exerting forces of any kind on each other. Consequently, no links can be established between them, as interference with each other is virtually non-existent.
However, the situation is pretty different in concentrated solutions, represented above on the right. As the number of molecules is increased, they are forced to come closer and interaction between them turns noticeable. The summation of intermolecular forces becomes a key factor, and the whole polymer mass no longer will show the same behavior it displayed when coils were surrounded by vast domains of solvent.
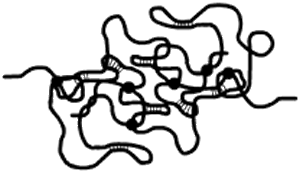
Therefore, the viscosity increases drastically, and the solution begins to exhibit a transition from a concentrated solution to a gel, because such coils will establish linkages at some points, constituting gradually an only giant crosslinked coil, called a macroscopic gel. Such macroscopic gel is pictured schematically below, showing the crosslinks as solid circles.

Have you asked yourself how these linkages between coils can be established? Surely there must be some kind of bond able to hold the molecules together. Well, indeed there is. Usually such molecules become linked in two ways: through secondary interactions (secondary valence gels) and by means of covalent bonds (covalent gels). Let's discuss each of them in detail.
Secondary interactions are reversible, and not only occur between different coils (intermolecular linkages), but also within the same coil (intramolecular linkages). Look at the pictures below. The one on the left shows a coil with intramolecular associations only. The picture on the right shows two coils with both intra and intermolecular linkages. The latter are represented by solid circles.
Normally, the formation of secondary valence gels depends on two factors: the strength of the intermolecular forces, which is a measure of the tendency of the polymer to establish non-covalent linkages, and the quality of the solvent.
Let's see. In the first place, it would help to understand what exactly
gelatin is made of. Did you know it's mainly protein collagen, i.e. a natural polymer? This means that there are
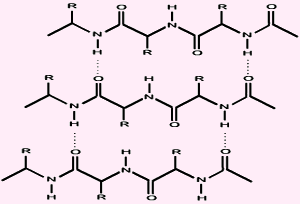
many amide groups in the backbone to establish bonds with each other. Those are not covalent bonds, of course, but a particular kind of reversible linkage called hydrogen bond. The picture below shows those non-covalent linkages between protein chains. Hydrogen bond is a strong interaction that however is usually broken upon warming. Think about water: you need to warm it up to 100�C if you want it to boil! That's the temperature at which the hydrogen bonds are broken, and the free water molecules switch from their liquid state to a gas state.
Other naturally occurring polymers that form gels through secondary
interactions include pectin, agar, and starch. That's why they are widely used in industry as thickeners, giving consistency to many foods.
However, PVC, not a natural polymer indeed, can
also form secondary valence gels if you choose an appropriate solvent. When PVC is mixed with such liquids called plasticizers (like phthalic, adipic, or sebacic acid esters for example) it constitutes thermo-reversible gels, commonly called "plasticized PVC", with rubbery or leathery consistency.
Up to now we've seen how secondary valence gels can be broken without difficulty when the secondary interactions that take place between coils are withdrawn. But can you break up covalent bonds in such an easy way?
Well, the answer is no. There isn't any solvent powerful enough to cause
the breaking of covalent bonds. Moreover, this type of linkage cannot be
broken even by heat. Remember crosslinked thermosets? If it could, there wouldn't be increasing interest in the development of thermoplastic elastomers, for example.
Thus covalent gels can be obtained in two ways: first, you can swell a
previously crosslinked polymer, choosing a suitable solvent. Want an
example? Here's one: polyacrylamide.
It's soluble in water, but crosslinked polyacrylamide is not; it can only be swollen in water.
Do you prefer to crosslink a functional dissolved polymer? Good. You can
also do this, as long as you add another bi or multi-functional compound
into the solution. If you're looking for some examples, then visit the epoxy resins or the diene polymers page.
So you now know what a gel is, and how many types of gels you can produce.
But surely you'll have many more interesting questions by now. For
example, being crosslinked materials, do gels show the same properties as,
say, elastomers or thermosets, i.e. solid materials? How can gels be characterized, anyway?
As in gels the coils remain fixed in space, not exchanging places as they do in concentrated solutions, the gel will not flow. That is an important property: imagine how it is for such products that are sold in tubes, such as toothpastes, cosmetics, or pharmaceuticals.
However, you might guess that the presence of the solvent within the reticular structure must have something to do with the behavior of such particular substances. And it has indeed: unlike solid molecules, each gel molecule could be considered as a dilute solution. This is a considerable advantage: it means that the coil segments can move rather freely, depending on the segment chemical structure, the concentration, the coil density, and obviously, the crosslinking degree. This movement is often called micro-brownian motion. That's why gels, under the influence of any deforming force, can react with an elastic retractive force. We then say gels have elasticity.
Like solid polymers, the strength of gels increases with the crosslinking degree, i.e. their hardness and toughness turn higher. The properties of the solid state are usually maintained when the system goes into gel. This means that brittle polymers will lead to brittle gels, and rubbery polymers will form gels with certain toughness.
But in a general sense, are gels mechanically powerful? Can they be compared to solid bodies? To find a reasonable answer, let's go back to the role that the solvent plays in such a system.
In solid polymers chains are close enough together. This means that the material can resist well any deformation without breaking up. But when solvent molecules come into play, as in gels, they penetrate among the chains, loosing them apart. Hence with separate chains acting as isolated entities, gels have no strength whatsoever to withstand a good deal of stress. Consequently, with some exceptions like plasticized PVC, we can conclude that gels, even the covalent ones, have poor mechanical properties most of the time.
The different properties shown by gels, depending on their crosslinking degree, might lead us to another interesting question: how can these properties be measured?
Well, as with solid polymers, in fact you can also evaluate mechanical
properties on gels, provided they have a moderate crosslinking degree. If
gels were not elastic enough, they would break before you could measure
such parameters like tensile
strength, elongation, and modulus. Or they would show a permanent deformation that would make difficult the quantitative evaluation of their mechanical properties.
However, if you are unable to measure mechanical properties for gels with a low crosslinking degree, you can establish another parameter which is characteristic of gels in particular: the swelling power.
What's that? In a physical sense, the swelling power gives us an idea about how much solvent can gels take up. Mathematically is a relationship between the weight (or volume) occupied by a swollen gel, W(s), and the weight (or volume) occupied by the corresponding dry gel, W(d). Of course, we can express all this by equations.
When considering weights, the equilibrium weight swelling ratio (qw) is given by:
If volumes are considered, then the equilibrium volume swelling ratio (qv) is defined by:
Doing a little more complex mathematical reasoning, you can also calculate the swelling degree percentage (%SD) given by:
The swelling power depends mostly on the solvent and the polymer chain
nature, and in polyelectrolytes (such as
polyacrylic acid) also depends on the degree of dissociation. However, that a gel is soft doesn't necessarily mean it should have a high swelling power. Many times, low swelling power is attributed to polymer chains with low molecular weights.
The swelling power has a great significance for gels. The more solvent the
gel takes up, the higher the swelling power is. That rings a bell.
Remember superabsorbers? If not, click here to find out. As their name suggests, superabsorbers like polyacrylamide, poly(vinyl alcohol), and poly(acrylic acid) can take up enormous amounts of water.
Gels like these mentioned above, with a remarkable ability to absorb water or aqueous solvents, usually more than 20% their own weight, are called hydrogels.
But you have many kinds of hydrogels in the market. There are porous and non-porous ones. Macroporous hydrogels that are able to dry and swell back without significant changes in their macroscopic structure are known as aerogels. On the other hand, dried non-porous hydrogels with capacity to absorb water by swelling are called xerogels.
While gels in general are widely used for a number of diverse applications, it's understandable that the interest for hydrogels is increasing constantly, particularly in their use as biomaterials.
From contact lenses and skin care uses to controlled drug release, application areas seem to be vast, especially with the advent of the so-called intelligent gels.
Since their discovery in 1975, intelligent gels have been tested in a number of different products. You might ask, for instance why gel soles are so comfortable. Well, that's because many of them are made using a viscoelastic gel that is soft at room temperature, but gets harder when making contact with the human body, i.e. it reacts with changes in temperature.
The ability to change their properties and the adherence to biological tissue is also another feature displayed by such hydrogels like poly(acrylic acid). This, coupled with the addition of some drugs into the gel matrix, makes them very useful for pharmaceutical applications like eye and nasal drops or sunscreens. That's because the gel becomes more viscous upon reaching the eye or nose, preventing the drops from dilution and washing by tears in the case of eye drops. Once inside the organ, the gel is capable to release the drug over a long period of time.
With sunscreens the procedure is different. Conventional sunscreens are usually greasy, so they're absorbed by the skin going directly into the bloodstream, where their utility is wasted. On the contrary, hydrogels are aqueous; they entrap the bioactive agent in micelles, so that it remains on the skin even longer.
Of course! Did you know that your muscles and internal organs are hydrogels? Sounds weird? It shouldn't... Just think, you have lots of proteins (i.e. natural polymers) in your body. But these proteins are surrounded by an enormous quantity of water. After all, about 80% of the animal body is water!
Now put those proteins in contact with water, and what's going on here? Well, we have a big hydrogel! All soft parts of our body are just a big gel, where continuous change in molecular size as well as crosslinking degree is taking place.
Although hydrogels play a key role in the new trends of biotechnology, gel
chemistry is not only circumscribed to organic molecules. In fact there
are many inorganic gels, obviously derived from inorganic polymers, particularly silicones and polysilanes. There is another important
branch of gel chemistry, named the sol-gel chemistry, which has been widely applied in the glass and ceramic fields, where research is still intense.
Commonly, the sol-gel process consists in the preparation of an inorganic network, working in solution at low temperatures. Such reaction involves three main steps: the formation of a colloidal suspension (called "sol"), the subsequent gelation of the sol in a continuous liquid phase ("gel"), and the removal of the solvent.
A great number of products are made by the sol-gel technology, such as fibers and composites, porous films, window insulators,
superconductors, fillers, and optical, dielectic and electronic coatings.
No surprisingly, the sol-gel process has even been applied for making high
purity silica for optical fibers.

If we consider the intermolecular forces, you might ask what type of polymers can form gels by means of secondary interactions when the solvent is water. Well, to find it out you could try an easy experiment in your kitchen.
 All you need is a small bowl and some gelatin previously stored in your refrigerator, like the delicious one that appears on the right. Now put a bit of gelatin into the bowl and warm it up a little. If you use a microwave, one minute is more than sufficient. Now look at the bowl. What happened? Gradually the gelatin melted and became a viscous liquid! But this is no surprisingly enough. Let the gelatin to cool down, and... it will solidify again! Why?
All you need is a small bowl and some gelatin previously stored in your refrigerator, like the delicious one that appears on the right. Now put a bit of gelatin into the bowl and warm it up a little. If you use a microwave, one minute is more than sufficient. Now look at the bowl. What happened? Gradually the gelatin melted and became a viscous liquid! But this is no surprisingly enough. Let the gelatin to cool down, and... it will solidify again! Why?
But what is an "appropriate" solvent when dealing with thermo-reversible secondary valence gels?
Just think about it. Usually poor solvents ensure an incomplete solvation, hence a solvation equilibrium exists. As molecules remain tightly coiled in the presence of such solvents, they mainly establish intramolecular rather than intermolecular linkages. The crosslinking density is therefore low, and the bonds established by secondary interactions are easily broken.
Covalent Gels
Mechanical Properties
Brittle polymers and gels include:
Rubbery polymers and gels include:
Poly(vinyl acetate)
Polyisobutylene Polystyrene
Polyacrylates Poly(vinyl carbazole)
Polyisoprene Poly(methyl methacrylate)
Polybutadiene copolymers
 Obviously, the strength will depend on the degree of crosslinking. The more crosslinked the material, the more elastic and stronger it is. You can check this easily. For example, all the gels you have for your personal care, such as bath, hair styling, even pain killers or other pharmaceutical gels, will completely break when you touch them with your fingers, like in the picture on the right. However, a mattress made of gels should be sufficiently elastic to make you feel comfortable, and strong enough so that it can stand your own weight without breaking when you lie down on it.
Obviously, the strength will depend on the degree of crosslinking. The more crosslinked the material, the more elastic and stronger it is. You can check this easily. For example, all the gels you have for your personal care, such as bath, hair styling, even pain killers or other pharmaceutical gels, will completely break when you touch them with your fingers, like in the picture on the right. However, a mattress made of gels should be sufficiently elastic to make you feel comfortable, and strong enough so that it can stand your own weight without breaking when you lie down on it.
Gel Characterization
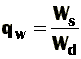
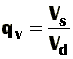
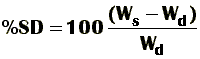
A WORLD OF GELS

Intelligent? Yes, they're smart enough because their swelling can be triggered by changes in such environmental factors like temperature, pH, solvent, ionic strength, light intensity, pressure, ultrasound irradiation, and magnetic or electric fields. Imagine that by varying the gel temperature in only 1�C, you can make the gel swell hundreds of times its own weight, or collapse, or even when it doesn't swell, it can display ability to change its physical properties. Neat, huh?
Checking The Intelligence Of "Intelligent Gels"
Gels In Your Body?
This Is Not The End

Return to Polymer Gels and Solutions

Return to Level Three Directory

Return to
Macrogalleria Directory
