
Elastomer is a big fancy word, and all it means is "rubber" or "stretchy" or even "bouncy." Some polymers which are elastomers include polyisoprene or natural rubber, polybutadiene, polyisobutylene, and polyurethanes. What makes elastomers special is the fact that they bounce, which is to say that they can be stretched to many times their original length, and can bounce back into their original shape without permanent deformation.
But why?
Putting Entropy to Work for You!
 Entropy is another fancy word for disorder or chaos. Things in our universe like entropy, and tend to become more disordered. That's why keeping your room messy is easier than keeping it neat. This dog is named Entropy, which is appropriate because she runs around like free-range chicken with its head cut off whenever her human lets her in the house. Polymer molecules are the same way. The molecules in a piece of rubber, any kind of rubber, have no order to them. They just wind and tangle around each other in one jumbled mess. They're perfectly happy this way.
Entropy is another fancy word for disorder or chaos. Things in our universe like entropy, and tend to become more disordered. That's why keeping your room messy is easier than keeping it neat. This dog is named Entropy, which is appropriate because she runs around like free-range chicken with its head cut off whenever her human lets her in the house. Polymer molecules are the same way. The molecules in a piece of rubber, any kind of rubber, have no order to them. They just wind and tangle around each other in one jumbled mess. They're perfectly happy this way.
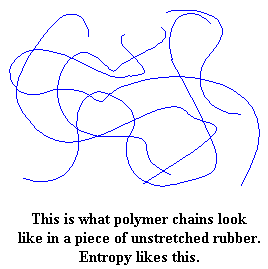
But now pull on the piece of rubber, and everything gets upset. The molecules are forced to line up in the direction in which the rubber is being pulled. When the molecules line up like this they become more ordered. If you stretch it far enough the chains will line up straight enough to crystallize. They don't like this. Remember, they like entropy (being disordered).
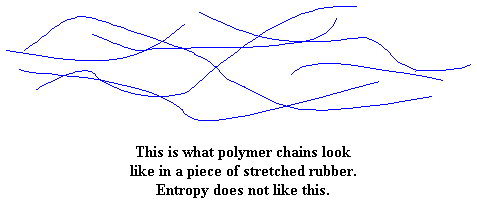
Now when you let go of this rubber sample you've been stretching, the molecules will quickly go back to their tangled and disordered state. They do this to return to a state of entropy. Remember, they like entropy. When this happens, the sample pops back to its original shape.
One Molecule Can Do a Lot
To help elastomers bounce back even better it helps to crosslink them. Crosslinking is the forming of links between the different polymer chains, joining them all into a single networked molecule. That's right, most objects made of rubber contain only one molecule! When the polymer chains are joined together like this, it is even harder to pull them out of their original positions, and so it bounces back even better when stretched.
But this makes elastomers hard to recycle. We usually recycle polymers by melting them down, but since melting is done by heating the molecules so that they flow past each other, there's no way to melt something that has only one molecule! Think about it. To make recyclable elastomers we need to find a way to tie the molecules together when the rubber is being used, but one which would allow the chains to separate when being heated and processed. The answer is called a thermoplastic elastomer.
A thermoplastic elastomer is just what the name sounds like, a rubber that acts like plastic. At normal temperatures it is durable and bouncy like most any other kind of rubber, but it can also be melted and easily formed into shapes when it is heated - like plastics.
One pretty common thermoplastic elastomer is SBS rubber which is used to make all the rubber parts on athletic shoes.
Thermoplastic Elastomers
 Many thermoplsatic elastomers are like this because they are copolymers made from two different polymers - one a rubber and the other a plastic. Because they can be melted, they are recyclable. But when they are cool they form crosslinks that are not permanent This makes then pretty rubbery.
Many thermoplsatic elastomers are like this because they are copolymers made from two different polymers - one a rubber and the other a plastic. Because they can be melted, they are recyclable. But when they are cool they form crosslinks that are not permanent This makes then pretty rubbery.

|
Return to Kinds of Polymers |

|
Return to Main Page |