

But just coming up with a few categories isn't enough for us. We have to come up with different classification systems. And the different systems can often be confused. So we're going to try to make it clear as possible by listing the different systems right up front. The first system is the Addition-Condensation system.
This system divides polymerizations into two kinds, and if you're
really clever you've probably figured out that those two kinds
of polymerization are
and
Now we call a polymerization an addition polymerization if the
entire monomer molecule becomes part of the polymer. We call
a polymerization a condensation polymerization if part of the
monomer molecule is kicked out when the monomer becomes part of
the polymer. The part that gets kicked out is usually a small
molecule like water, or HCl gas. Let's look at some examples
to illustrate the point.
When ethylene is polymerized to make polyethylene,
the every atom of the ethylene molecule becomes part of the polymer.
The monomer is added to the polymer outright. You might
say that an addition polymer is like a good friend who accepts
everything about you, the pleasant and the unpleasant alike.
But a condensation polymer is more like a snotty social club that
says, "Sure you can join, but only if you ditch those friends
of yours". You see, in a condensation polymerization, some
atoms of the monomer don't end up in the polymer. When
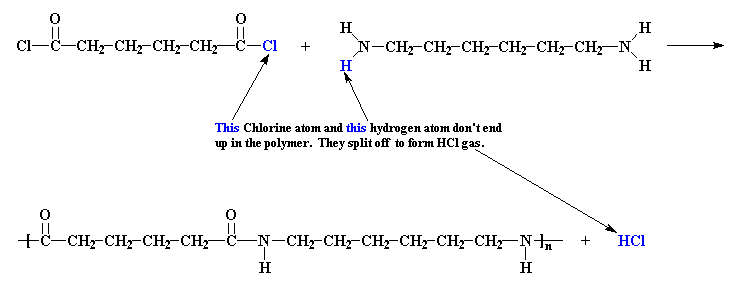
nylon
6,6 is made from adipoyl chloride and hexamethylene diamine, the
chlorine atoms from the adipoyl chloride, each along with one
of the amine hydrogen atoms, are expelled in the form of HCl gas.
Because there is less mass in the polymer than in the original
monomers, we say that the polymer is condensed with regard
to the monomers. The byproduct, whether it is HCl gas, water, or
whatnot, is called a condensate.
The Bottom Line
Want the short version? Condensation polymerizations give off
byproducts. Addition polymerizations don't.
Now let's talk about the next system of classification: The Chain Growth-Step Growth system.
The other system of classifying polymerizations again divides
polymerization reactions into two categories, and those are, all together
now:
and
Very good. Now the differences between a chain growth polymerization
and a step growth polymerization are a little more complicated than the
differences between an addition polymerization and a condensation
polymerization. It goes something like this:
In a chain growth polymerization, monomers become part of the polymer one
at a time. Pretty simple, huh? To show you how it works we've got a
picture of a chain growth polymerization here, namely the
anionic polymerization of styrene to make
polystyrene. Take a look at it.
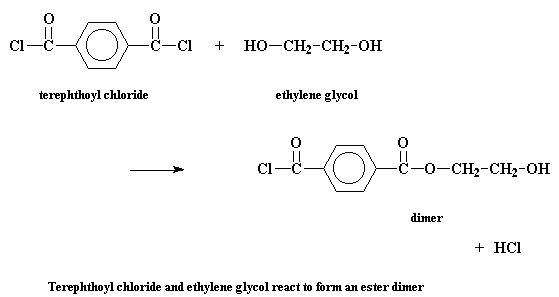
But in step growth polymerization, things are more complicated. Let's
take a look at the step growth polymerization of two monomers, terephthoyl
chloride and ethylene glycol, to make a polyester
called
poly(ethylene terephthalate). The first thing that happens is that the
two monomers will react to form a dimer. Sounds simple enough.
Now at this point in a chain growth system, only one thing could happen:
a third monomer would add to the dimer to form a trimer, then a fourth
to form a tetramer, and so on. But here, in the free land of step growth
polymerization, that dimer can do a lot of different things. It can of
course react with one of the monomers to form a trimer:
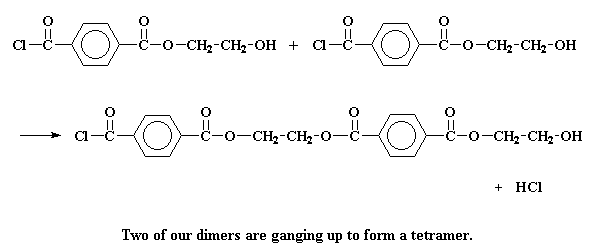
But it can do other things, too. It may react with another dimer to form a
tetramer:
Or it may even get really crazy react with a trimer to form a pentamer.
Making things more complicated, these tetramers and pentamers can react
to form even bigger oligomers. And so they grow and grow until
eventually the oligomers are big enough to become polymers.
Just think of the way your bank keeps merging with other banks and getting
bigger and bigger, and you'll get the idea.
The Bottom Line
The main difference is this: In a step growth reaction, the growing
chains may react with each other to form even longer chains. This
applies to chains of all lengths. The monmomer or dimer may react in
just the same way as a chain hundreds of monomer units long. But in an
addition polymerization, only monomers may react with growing chains.
Two growing chains can't join together the way they can in a step growth
polymerization.
Now I'm sure a lot of you bright people out there noticed that our step
growth polymerization to make a polyester produced a byproduct, HCl gas.
This of course will make it condensation polymerization as well as a
step growth polymerization.
You may have also noticed that our chain growth polymerization of
styrene did not produce a byproduct. Yes, this is an addition
polymerization in addition to being a chain growth polymerization.
It's easy to conclude here that a step-growth polymerization and
condensation polymerization are the same thing, and that a chain growth
polymerization and an addition polymerization are the same thing. But
this just isn't true. There are addition polymerizations which
are step growth polymerizations. One example is the polymerization which produces polyurethanes.
There are also
condensation polymerizations
which are chain growth polymerizations. Trying to reconcile the chain
growth-step growth classification system and the addition-condensation
classification system is really a waste of time. Each has its own
criteria, and the distinctions made by one system are not always going to be
the same as the distinctions made by the other.
So don't try to reconcile the two systems. Just know that polymerizations
can be step growth or chain growth, and they can be condensation or
addition.
The Addition-Condensation System
Condensation Polymerization
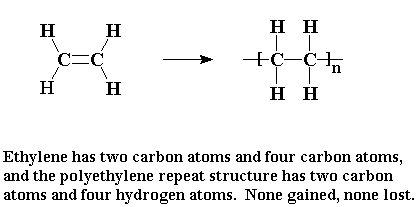
The Chain Growth-Step Growth System
Chain Growth Polymerizations
Step Growth Polymerizations

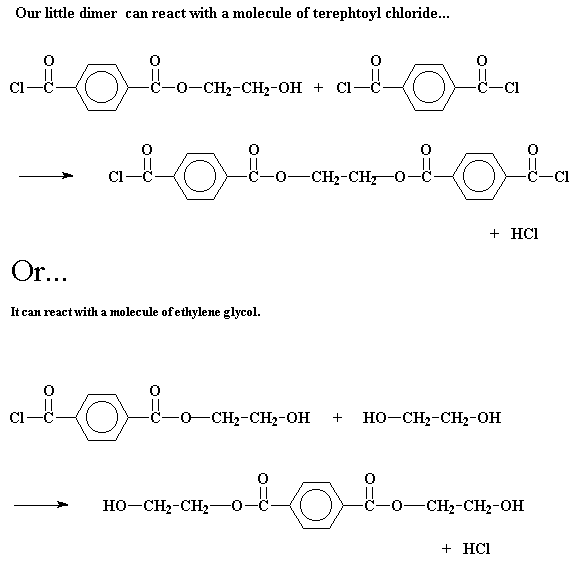
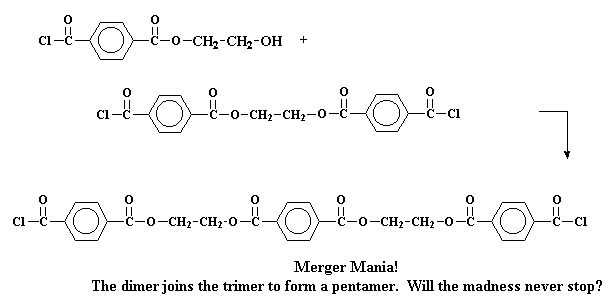
Irreconcilable Differences
 Return to Level Four Directory
Return to Level Four Directory
 Return to
Macrogalleria Directory
Return to
Macrogalleria Directory