

Also known as
PTFE or Teflon®!
Chances are you've never heard of poly-tetra-fluoro-ethylene, but I'll bet you've heard of Teflon®, the stuff used on nonstick pans to make them nonstick. The name Teflon® is a trade name (from DuPont) for this polymer, but sometimes it just goes by its initials, "PTFE".
What can PTFE do?
Teflon® is used to make non-stick cooking pans, and anything else that needs to be slippery or non-stick. PTFE is also used to treat carpets and fabrics to make them stain resistant. What's more, it's also very useful in medical applications. Because human bodies rarely reject it, it can be used for making artificial body parts.
Did you know that Teflon® is also made into "Thread Seal Tape" used by plumbers - or anybody who fixes leaky faucets or changes a shower head? Before screwing two metal pipes together, they wrap a piece of this tape around the threads first. It stretches a little and makes a great seal - much better than metal-on-metal. And, since it's slippery, the pipes are easier to screw together - no more scritchy scrapiness.
You can find Teflon® tape in any store that sells plumbing supplies - most hardware stores will have it, but they might call it by its real name, PTFE.
Activity: Write a Secret Message
PTFE Up Close
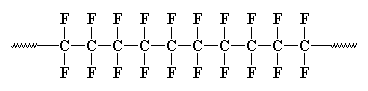
Polytetrafluoroethylene, or PTFE, has a carbon backbone chain, and
each carbon has two fluorine atoms attached to it. Part of a chain looks like this, but picture the chain being thousands of carbon atoms long:
And for those of you who are wondering, the monomer tetrafluoroethylene looks like this:
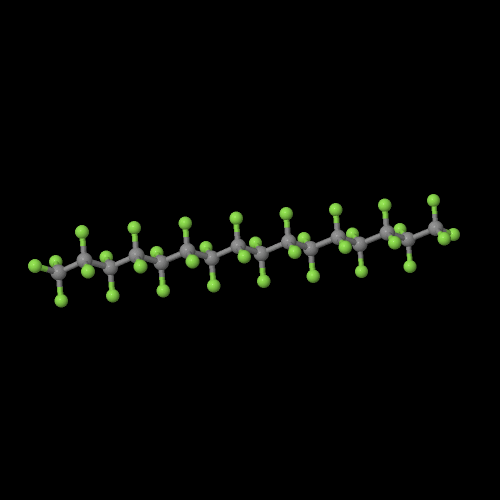
 Click on the monomer image to pop up a 3D version you can examine in detail. Do the same for the polymer image on the right so you can see the difference.
Click on the monomer image to pop up a 3D version you can examine in detail. Do the same for the polymer image on the right so you can see the difference.
How to be Slippery
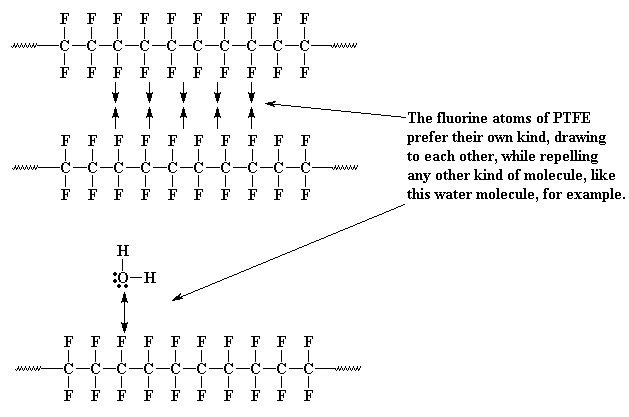
Fluorine is a very strange element. When it's part of a molecule, it doesn't like to be around other molecules, even the fluorine atoms on other molecules. But it likes other kinds of molecules even less. So a molecule of PTFE, being just chock full of fluorine atoms as it is, would like to be as far away from other molecules as it can get. For this reason, the molecules at the surface of a piece of PTFE will repel the molecules of just about anything that tries to come close to it. That's why nothing sticks to PTFE.
Because it's non-stick, PTFE means you can fry things without grease or butter. This means less fat and cholesterol, for a healthier heart.
PTFE has other interesting properties. First of all, it doesn't melt so that you can mold it. Instead, you take a powder form of the polymer right from the reactor you made it in, layer that on what you want to coat, and apply heat and pressure. This process is called "sintering," and is how most PTFE coated fry pans are made.
Now for the reason that sintering works. Turns out that fluorine atoms, even those on a polymer chain like in PTFE, don't really like to be near each other. The chains repel each other similar to how they repel almost everything else. So even though PTFE feels like a solid, all the polymer chains inside are wiggling around, trying to get as far away from their neighbors as possible. This means that if you press on it hard, especially with some heat applied, the whole mess of polymer chains will wiggle around, reorient and take on a new shape.
How to be Inert
PTFE is more than just slippery. It's also useful because it won't react with anything. Why, you ask? First of all, if it repels everything, then no molecule can get near it to react with it!
Then there is the fact that the bond between the fluorine atom and the carbon atom is just really, really strong. It's so stable that nothing will react with it, even when it gets as hot as a frying pan!
Oops! Hey, it's a polymer!!
Polytetraflouroethylene is another of those amazing accidental discoveries of science. In the late 1930s, when PTFE was discovered in DuPont's laboratories, DuPont was not at all concerned with nonstick frying pans or artificial heart valves. What they were really interested in was refrigeration. At the time, refrigerators used things like ammonia and sulfur dioxide as refrigerants. These are pretty nasty things to have leaking out of your refrigerator and into your kitchen. The quest was on, then, to make a non-toxic refrigerant. One of the compounds being investigated was tetrafluoroethylene.
One chemist at DuPont who was working on the project was named Roy Plunkett. One day Roy Plunkett opened up a brand new tank of tetrafluoroethylene gas, and nothing came out! He weighed it, and sure enough it was full. So he sawed the tank open and found a white powder where the gas was supposed to be. That powder, of course, was PTFE that had polymerized from tetrafluoroethylene gas.
This is the kind of accident that makes science fun. There are kinds of accidents in science which aren't fun, say, those which involve big booms, but we won't talk about those right now. The fun kind of accident, which all scientists hope for, is an unexpected discovery which opens up a whole new area of investigation and makes you famous.
And then someday you might get your name mentioned in an educational website.
Making PTFE
The monomer is tetrafluoroethylene - just like ethylene, but with fluorine atoms instead of hydrogen.

Source: Roberts, Royston M.; Serendipity: Accidental Discoveries in Science; John Wiley and Sons; New York; 1989.

|
Return to Kinds of Polymers |

|
Return to Main Page |