
Glossary

Anion - an atom or molecule which has a negative electrical charge. (see: ion)
Cation - an atom or molecule which has a positive electrical charge. (see: ion)
Complex - two or more molecules which are associated together by some type of interaction of electrons, other than a covalent bond. (see: covalent bond)
Copolymer - A polymer made from more than one kind of monomer. (see: monomer)
Covalent bond - a joining of two atoms when the two share a pair of electrons.
Crosslinking - crosslinking is when individual polymer chains are linked together by covalent bonds to form one giant molecule. (see: elastomer, thermoset)
Crystal - a mass of molecules arranged in a neat and orderly fashion. In polymer crystal the chains are lined up neatly like new pencils in a package. They are also bound together tightly by secondary interactions. (see: secondary interactions)
Electrolyte - a molecule that separates into a cation and an anion when its dissolved in a solvent, usually water. For example, salt, NaCl separates into Na+ and Cl- in water:
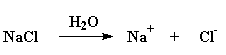
Emulsion - a mixture in which two immiscible substances, like oil and water, stay mixed together thanks to a third substance called an emulsifier. The emulsifier is usually something like a soap, whose molecules have a water-soluble end and an organic-soluble end. The soap molecules form little balls called micelles, in which the water-soluble ends point out into the water, and the organic-soluble ends point into the inside of the ball. The oil is stabilized in the water by hiding in the center of the micelle. Thus the water and oil stay mixed.
Entropy - disorder. Entropy is a measure of the disorder of a system.
First order transition - a thermal transition that involves both a latent heat and a change in the heat capacity of the material. (see: heat capacity, latent heat, second order transition, thermal transition)
Free radical - an atom or molecule which has at least one electron which is not paired with another electron.
Gel - a crosslinked polymer which has absorbed a large amount of solvent. Crosslinked polymers usually swell a good deal when they absorb solvents. (see: crosslinking)
Glass transition temperature - the temperature at which a polymer changes from hard and brittle to soft and pliable.
Heat capacity - the amount of heat it takes to raise the temperature of one gram of a material one degree Celsius.
Hydrodynamic volume - the volume of a polymer coil when it is in solution. This can vary for a polymer depending on how well it interacts with the solvent, and the polymer's molecular weight.
Hydrogen bond - a very strong attraction between a hydrogen atom which is attached to an electronegative atom, and an electronegative atom which is usually on another molecule. For example, the hydrogen atoms on one water molecule are very strongly attracted to the oxygen atoms on another water molecule.
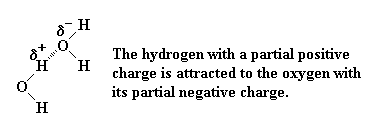
Ion - an atom or molecule which has a positive or a negative electrical charge.
Latent heat - the heat given off or absorbed when a material melts or freezes, or boils or condenses. For example, when ice is heated, once the temperature reaches 0 oC, its temperature won't increase until all the ice is melted. The ice has to absorb heat in order to melt. But even though it's absorbing heat, its temperature stays the same until all the ice has melted. The heat required to melt the ice is called the latent heat. The water will give off the same amount of latent heat when you freeze it.
Le Chatelier's principle - this principle states that if a system is placed under stress, it will act so as to relieve the stress. Applied to chemical reactions, it means that if product or byproduct is removed from the system, the equilibrium will be upset, and the reaction will produce more product to make up for the loss. In polymerizations, this trick is used to make polymerization reactions reach high conversion.
Ligand - an atom or group of atoms which is associated with a metal atom in a complex. Ligands may be neutral or they may be ions. (see: complex, ion)
Matrix - in a fiber reinforced composite, the matrix is the material in which the fiber is embedded, the material that the fiber reinforces. It comes from a Latin word which means "mother", interestingly enough.
Modulus - the ability of a sample of a material to resist deformation. Modulus is usually expressed as the ratio of stress exerted on the sample to the amount of deformation. For example, tensile modulus is the ration of stress applied to the elongation which results from the stress. (see: elongation, stress)
Olefin Metathesis - a reaction between to molecules, both
containing carbon-carbon double bonds. In olefin metathesis, the double
bond carbon atoms change partners, to create two new molecules, both
containing carbon-carbon double bonds.

Oligomer - a polymer whose molecular weight is too low to really be considered a polymer. Oligomers have molecular weights in the hundreds, but polymers have molecular weights in the thousands or higher.
Plasticizer - a small molecule that's added to polymer to lower its glass transition temperature. (see: glass transition temperature).
Ring-opening polymerization - a polymerization in which cyclic monomer is converted into a polymer which does not contain rings. The monomer rings are opened up and stretched out in the polymer chain, like this:
Secondary interaction - interaction between two atoms or molecules other than a covalent bond. Secondary interactions include hydrogen bonding, ionic interaction, and dispersion forces. (see: hydrogen bond)
Second order transition - a thermal transition that involves a change in heat capacity, but does not have a latent heat. The glass transition is a second order transition. (see: first order transition, glass transition temperature, heat capacity, latent heat, thermal transition)
Strain - the amount of deformation a sample undergoes when one puts it under stress. Strain can be elongation, bending, compression, or any other type of deformation. (see: elongation, stress)
Strength - the amount of stress an object can receive before it breaks. (see: stress)
Termination - in a chain growth polymerization, the reaction that causes the growing chain to stop growing. Termination reactions are reactions in which none of the products may react to make a polymer grow.
Thermoplastic - a material that can be molded and shaped when it's heated.
Thermal transition - a change that takes place in a material when you heat it or cool it. Examples of thermal transitions include melting, crystallization, or the glass transition. (see: glass transition temperature)
Thermoset - a hard and stiff crosslinked material that does not soften or become moldable when heated. (Compare thermosets with thermoplastics, which do become moldable when heated.) Also, thermosets are different from crosslinked elastomers. Thermosets are stiff and don't stretch the way that elastomers do. (see: elastomer, thermoplastic)
Toughness - a measure of the ability of a sample to absorb mechanical energy without breaking, usually defined as the area underneath a stress-strain curve. (see: stress, strain)

|
Return to Macrogalleria Directory |
