Isotactic Poly(vinyl ether)
Something Made A Long Time Ago,
But Now Made a New Way...
Use your mouse on the model to rotate and zoom.
Click items below for other modifications.
spin on spin off
spacefill wire thick wire ball&stick
dots: Vanderwaals dots off
Look closely at the model above. Click on it, move it around some, then zoom in so it's really big. Rotate this enlarged view around and you should begin to see the helical shape of the interior part of the model shown. This structure has enormous importance in understanding the solid state properties of this wonder material. "Wonder material?" you ask? Sure, but right now it's the "look at it and wonder why..." kind of wonder. Let's talk more.
"Make it as simple as possible, but no simpler-" Albert Einstein
with the corollary that "simple" doesn't always work.
So let's first talk about cationic polymerizations in general. We'll use isobutylene polymerization as our example. "Why?" you ask, "It's not even a vinyl ether." The answer is simple: its cationic polymerization is one of the few that are efficient and give a usable product. Let's dig into the mechanism more.
When a carbocation of some kind sees a vinyl double bond, it senses a whole bunch of electron density there that it would love to share. Kind of like when my dog watches me eating steak: "Let's share!"
So if cations can't share with a neighbor atom, they really, really want to bond with something else with lots of negative charge density. So that part is good. "And," you ask, "Just what gets the whole thing started in the first place?" Well, of course, that would be an initiator, which is usually a carbocation made for that purpose, but protons work as well sometimes, under the right conditions.
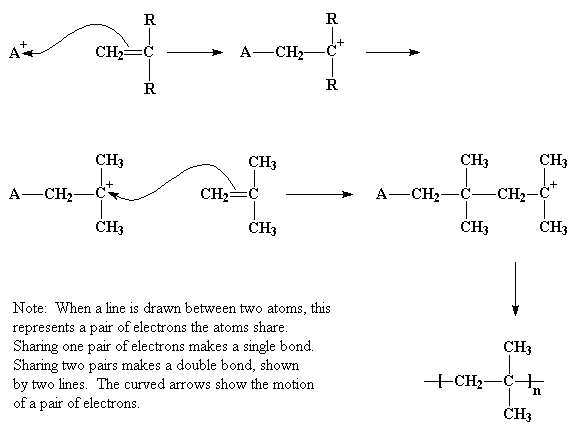
Now for the next step, the repetitive addition of the cation to the terminal carbon (see above). In essence, the incoming cation is hungry. So hungry in fact, that it takes ALL of the electron density in the double bond for itself. That makes for a new bond with two electrons in it, satisfying that insatiable hunger. But wait! What about the other carbon, the one left all alone when its one electron (that it so graciously shared in the double bond) is ripped away to form the new bond? Not a happy camper! It's left with a full positive charge, which means it's missing a pair of electrons in its outer shell. It now only has 3 pairs (six electrons) which makes it re-hybridize to a flat, trigonal planar structure which is really no use to anyone for anything, especially if you're trying for stereoregular polymer.
So what does it do? Why it turns around and steals electron density from its nearest neighbors, in this case, a pair of methyl groups. That helps a lot, makes it feel better and hey, what are those methyls going to do about it anyway?
Now here's the reason cationic polymerization doesn't work for most vinyl monomers. First, there's not enough electron density lying around to make it happen. And second, even if it does, and you form a new carbocation from a vinyl bond, all kinds of other reactions take place instead of propagation. These can include inter- and intramolecular hydride or carbanion shifts, and termination through combination of the cation with whatever counterion is present. In short, it's normally hard to control, hard to get high molecular polymer with, and definitely doesn't give stereoregular product.
"Not sure I get that last part..." you state in crinkle-brow puzzlement. Forgot to mention this: because carbocations are so reactive, they do whatever they're going to do really, really fast. If for some strange reason propagation does occur, it occurs so fast from that non-chiral trigonal planar intermediate that there's no time to make a regular sequence. In short, no stereoregular backbone of any kind.
Oh, and that leads to another problem: reaction exotherm. You should know by now that vinyl polymerization is usually a down-hill process. The polymer is more thermodynamically stable than the collection of monomers. And that means heat is liberated during polymerization. Normally, at speeds occurring for radical and anionic polymerizations, we can control that heat by cooling or using solvent. But here, the reaction is so incredibly fast that, no matter how hard we try, the local temperature around the active chain end is hard to control. And with the higher temperature generated, any chance of sterically controlled or counterion induced stereo-sequence induction is lost in the wind (so to speak).
And just to wrap up the polyisobutylene example, you might ask, "So why does it work there?" Answer is simple: it's because nothing much else can happen. First, the tertiary carbocation intermediate formed is pretty stable, not great but not so bad. It's got two methyl groups attached, and they donate enough electron density to calm the cation down some. As far as rearrangement goes, a 1,2-hydride shift would give you a primary carbocation on a methyl carbon, and that just doesn't happen. So if you can keep the temperature under control, you can make pretty usable PIB.
Now for the weird part. With such a perfectly regular structure, with two pendent methyl groups per repeat unit, you might expect polyisobutylene to be crystalline with a fairly high melting point. Not so. In fact, it's an elastomer at room temperature, which is neat. Even more interesting, and more useful, is the fact that the high symmetry allows close packing of the chains even though they don't crystallize. And THAT means good barrier properties to gas transport. And THAT is why PIB (as it's affectionately known) is used in tire inner tubes and the inner liner of tubeless tires: it keeps the air in so your tires don't run flat too fast. For more on PIB, go here.
NOW for the "rest of the story..."
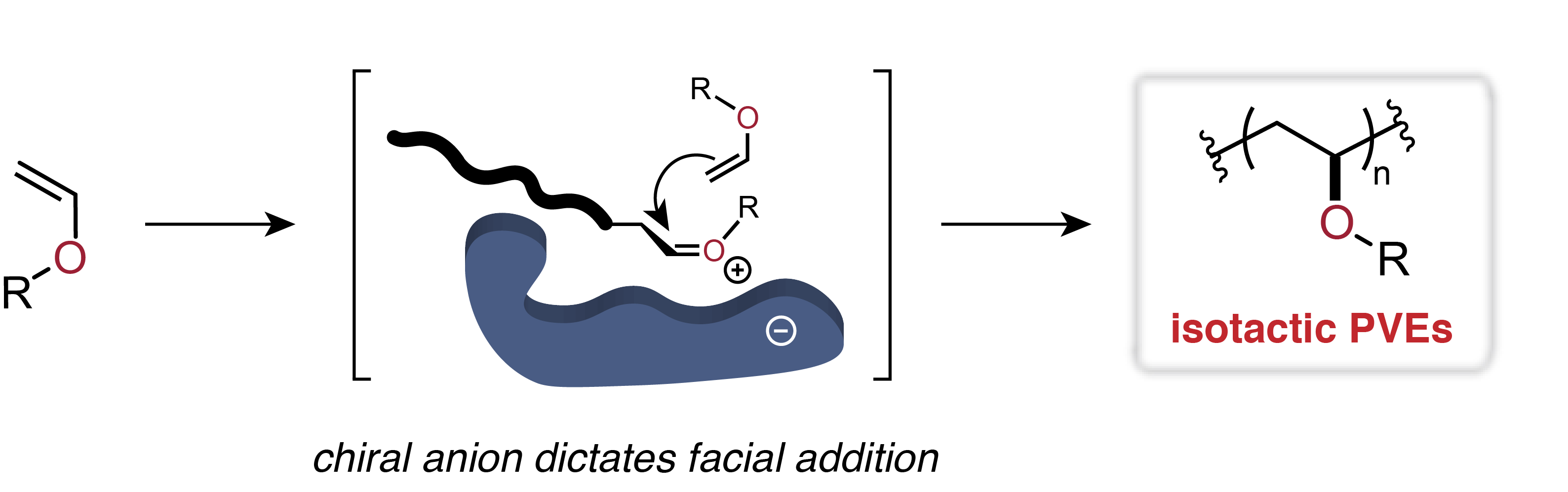
Ok, then, it's time to talk about polymerization of vinyl ethers. Now a propagating carbocation here would be located on the carbon next to the ether oxygen. You might think to yourself, "So what? Oxygen is more electronegative than carbon, so how does it help?" Great question, and yes, oxygen is more electronegative than neutral carbon, but not a carbon with a positive charge on it. More important is the fact that the ether has not one, but two, pairs of electrons just hanging around in its outer shell. Once that carbocation starts to form, it sucks those elctrons in to help stabilize it. Well, hey, that lazy oxygen wasn't doing anything with its lone pairs anyway... And how's that old saying go? "When push comes to shove ..." and substitute "pull" for "push."
So that leads to the stabilized ionic form of the cation next to a vinyl ether oxygen, one in which one of the oxygen atom's lone pairs is pulled over to form a double bond with the attached carbon (see below). It's not really that simple, but good enough for this discussion. And you need to know that the oxygen has rehybridized from tetrahedral to trigonal planar. But now everyone's happy electronically, right? Well, happy enough to allow somewhat controlled vinyl polymerization but not to give stereoregular polymer. What to do? What to do?

One thing polymer chemists figured out back in the dark days of polymer synthesis was to use temperature to help. Remember I said this vinyl polymerization was exothermic with heat liberated raising the local temperature? Why not cool the whole mess BEFORE you start the polymerization? Maybe that would help? In fact it does and you can make pretty darn good polymer by lowering the temperature to dry ice levels before you add the initiator. By the way, the dry ice/acetone bath used stays at -78 oC for the duration of polymerization, which is really, really cold.
Tested isobutyl vinyl ether synthesis
To make a highly crystalline sample the way it was first made this way, we have a procedure for you to follow. It uses Dry Ice temperatures and liquid propane solvent with BF3 as catalyst/initiator. An interesting feature of this method is that it can be used to make a variety of isotactic poly(vinyl ethers). So if you want to see one of those earlier procedures, one that's "tried and true," why here's a link to a tested procedure for making highly isotactic poly(isobutyl vinyl ether). And click here to download a copy of your very own.
What about the counterion??
It turns out, as is the case so often in science, that's there's more than one way to skin a cat. Ok, sorry for the analogy. I really don't hate cats or try to remove their outer layers in any way. The point is that if you want stereoregular poly(vinyl ethers), than there actually is a different way then just lowering the temperature. All else being said, though, that still plays a major role.
So what is this new addition to our toolbox of cationic polymerization methods? Simply this: the counter ion, that negatively charged species that interacts with and neutralizes the propagating carbocation. And THAT, my friends, is where the new development shines. The figure below pretty well sums up the discovery that has led to an augmented way to make isotactic poly(vinyl ether).
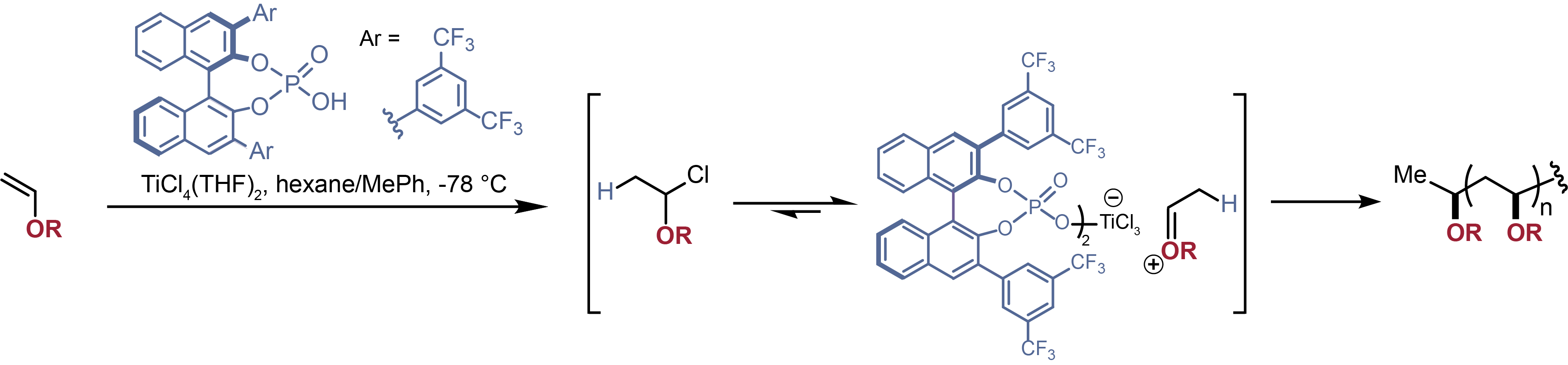
But that's not the whole story, of course. If you look more closely and use a trick to slow down the usual rapid-fire propagation step, you get much better control of the complex and better formation of stereosequences. So here's the details in all their glory:
So what about physical properties? Is this new stuff any better than the gooey blobs made the old way? You bet! Take a look at the figure on the left below: solid versus viscous liquid. Not good enough for you? Take a gander at the tensile test image: the isotatic poly(isobutyl vinyl ether) keeps its solid form, stretches beautifully to several times its original length.

So this month's "POTM" is unique for several reasons, starting with the finely-tuned synthesis conditions using a specific kind of counter ion plus reversible addition-elimination of the halide, all the way to final physical properties which may make it commercially useful. "So is it on the market yet?" you ask. Nope, but at least a patent has been applied for...

To learn more about recent advances and historical perspective on poly(vinyl ethers), and to understand how to use polymerization "tricks" to generate high molecular weight material, click here.
And now to recognize the actual inventors of this new form of an old polymer, and give them a grateful "Thank You!" for the beautiful images they provided:
Teator, A. J. & Leibfarth, F. A. A catalyst-controlled stereoselective polymerization of vinyl ethers to form polar thermoplastics. Science 2019, 363, 1439; weblink at: http://dx.doi.org/10.1126/science.aaw1703
References on poly(vinyl ethers), to name just a few:
G. Schroeder, in Ullmann's Encyclopedia of Industrial Chemistry (Wiley, 2000), vol. 28, pp. 481-485.
L. Fishbein, B. F. Crowe, Makromol. Chem. 48, 221-228 (1961).
M. Ouchi, M. Kamigaito, M. Sawamoto, Macromolecules 32, 6407-6411 (1999).