Polystyrene is one of those polymers that's all over the place. Polystyrene is an inexpensive and hard plastic, and probably only polyethylene is more common in your everyday life. The outside housing of the computer you're using now is probably made of polystyrene, as well as the housings of things like hairdryers, TVs and kitchen appliances. Model cars and airplanes are made from polystyrene, as well as many other toys. There's also foam packaging and insulation, and a lot of the molded parts on the inside of your car, like the radio knobs.
| Polystyrene is also used to make drinking cups - the hard plastic ones and also the soft foamy ones. A popular brand of polystyrene foam is called StyrofoamTM. |

|
Would you believe all three of these cups are made from polystyrene?! |
|
Here's a molecule of polystyrene. You can see it in this 3-D model on the right or in the diagram below. Click the 3-D picture to see an interactive version.
 What, you guys again?! |

|
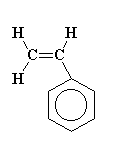
Polystyrene chains are made from many styrene molecules. (So, the monomer is styrene.) Here you can see a model and a diagram of the styrene monomer. Go ahead, click on the image on the left and play with the 3D model that pops up! This is really how the molecule looks at the nano-scale.

Polystyrene's Personalities
Most polystyrene you find will be the ordinary stuff used in CD cases and plastic forks. It's brittle and breaks easily. But there are also newer kinds which are stronger and can be used for special jobs. Let's talk about some of those.
Polystyrene that's the same, but different....
There's a new kind of polystyrene out there, called syndiotactic polystyrene. Now, to understand how this is different from plain old polystyrene, we have to picture the polymer chain all stretched out, like this:
This doesn't show the whole picture, though. When we look at it in 3D, that big blobby pendant group can be sticking out toward you (like it's in front of the computer screen), or on the other side (like it's behind the screen), kind of like this:
In plain (or atactic) polystyrene, there's no regular order to which side of the chain those pendant groups are on, just like the structure shown above.
| In syndiotactic polystyrene, every other pendant group is sticking out at you, and the other ones are towards the back, away from you. (By the way, syndiotactic sounds like: sinn-dee-oh-tack'-tick.) |

|
Here you can see the new syndiotactic polystyrene alongside the old atactic polystyrene. Again, click on the images to pop up versions you can twist and turn, zoom in and zoom out, to really get a feel for what these two polymers look like.

|

|
Notice how straight and regular the syndiotactic molecule is, compared to how bunchy and "disorganized" the normal polystyrene looks. The new syndiotactic polystyrene is crystalline (that's just another way of saying "it's all ordered and organized"), which makes it stronger and more resistant to heat and chemicals. It costs more to make, so it's not used to make cups and plastic knives. Scientists feel like it would be useful to make medical equipment, because its tough crystalline structure makes it able to resist the moisture, heat and cleaners used to sterilize it.
Go Ahead, Hit It Hard!
But there are still some fun things you can do with old-fashioned atactic polystyrene. Wanna see something really nifty?

What would happen if we were to take some styrene monomer, and we put some polybutadiene rubber in the mix? Take a look at polybutadiene, and you'll see that it has double bonds in it that can polymerize. We end up with the polybutadiene forming a graft copolymer with the styrene monomer. A graft copolymer has one kind of polymer for the backbone chain, with chains growing out of it that are made from a different kind of polymer. In this case, it's a polystyrene chain with polybutadiene chains growing out of it.
These rubbery chains hanging off of the backbone chain do some good things for polystyrene. They act to absorb energy when the polymer gets hit with something. This makes it stronger, not as brittle, and capable of taking harder impacts without breaking than regular polystyrene. This material is called high-impact polystyrene, or HIPS for short.
Another thing that happens when polystyrene and polybutadiene get mixed is that there are still some separate chains of each polymer that form what's called a polymer blend. But it's the polystyrene chains with the rubber attached that give HIPS its good qualities.
SBS
 Polystyrene is also part of a type of hard rubber called
poly(styrene-butadiene-styrene), or SBS rubber.
SBS rubber is a thermoplastic elastomer. That means it acts like plastic and rubber at the same time.
Polystyrene is also part of a type of hard rubber called
poly(styrene-butadiene-styrene), or SBS rubber.
SBS rubber is a thermoplastic elastomer. That means it acts like plastic and rubber at the same time.
This stuff is good for things that are made in molds (like plastics), but still need to be rubbery. A good example is the bottoms of running shoes. Most of these are SBS rubber.
|
Other polymers used as plastics include: |

NOW PLAYING at the Macroplex Cinema! |

|
Return to Kinds of Polymers |

|
Return to Main Page |

 The difference between polystyrene and polyethylene is that each repeat unit in styrene has a big
The difference between polystyrene and polyethylene is that each repeat unit in styrene has a big