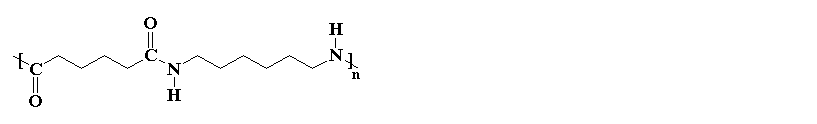
Nylons are one of the most common polymers used as a fiber. Nylon is found in clothing of many kinds today. In fact, its fibers are very similar to the natural polymer silk, which explains one of its earliest uses. It's first commercial success was as a replacement for silk in women's stockings because silk was expensive and hard to get. This was especially important in 1940 when this all happened. The new stockings were very popular, but soon the U.S. entered World War II, and all that nylon was needed for the war - to make parachutes and rope. Nowadays there's plenty of nylon to go around... and plenty of silk for that matter.
Nylon is also used in other places, such as in the form of a thermoplastic. Before stockings or parachutes, the very first nylon product was a toothbrush with nylon bristles.
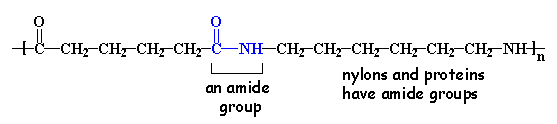
If you look at the nylon diagram above you'll see some atoms in blue. That segment is called an amide group. Because of that functional group in the polymer chains, nylons are called polyamides. The amide groups link the repeat units (from the two monomers) in the backbone chain.
Proteins, such as the silk that nylon was made to replace, are also polyamides. They're natural polymers made using enzymes that function in water, unlike the way we make nylons in huge kettles at high temperatures.
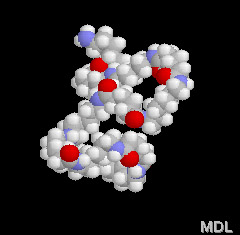
These amide groups are very polar and can hydrogen bond with each other. Because of this, and because the nylon backbone is so regular and symmetrical, nylons make great fibers because they are crystalline - which means the molecules all lock together in a regular pattern. It is easy to make out the nylon chain and it's pattern in the 3-D model on the right. Spin it around with your mouse and check it out!
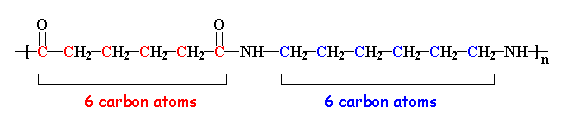
The nylon in the pictures on this page is called nylon 6,6, because each repeat unit of the polymer chain has two stretches of carbon atoms; each is six carbon atoms long. It is generically called an "A-A/B-B" polymer. Other nylons can have different numbers of carbon atoms in these stretches. Oh, and it's important to note that the two numbers in a nylons name refer to the diamine (first number) and the diacid (second one).
 Another kind of nylon is nylon 6. It's a lot like nylon 6,6 except that it only has one kind of carbon chain, which is six atoms long. In the generic naming convention, nylon 6 is an "A-B" polymer. The "A" refers to one of the functional groups (the amine, if you like) and the "B" refers to the other (in this case, the carboxylic acid).
Another kind of nylon is nylon 6. It's a lot like nylon 6,6 except that it only has one kind of carbon chain, which is six atoms long. In the generic naming convention, nylon 6 is an "A-B" polymer. The "A" refers to one of the functional groups (the amine, if you like) and the "B" refers to the other (in this case, the carboxylic acid).

Nylon 6 doesn't behave much differently from nylon 6,6, although it does have a melting point that's about 40 C lower. The only reason both are made is because DuPont patented nylon 6,6, so other companies had to invent nylon 6 in order to get in on the nylon business. It worked, too, since there are now huge markets for both.
Aramids

NOW PLAYING |
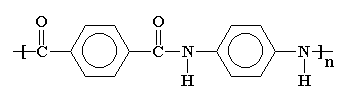
Aramids are a family of nylons including Nomex® and Kevlar® that are made with aromatic monomers. The structure above shows two of those monomer units, one an aromatic diamine and the other an aromatic diacid. Kevlar® has such outstanding properties that it is used to make things like bulletproof vests and puncture resistant bicycle tires. I suppose one could even make bulletproof bicycle tires from Kevlar®, if you felt the need.
Blends of Nomex® and Kevlar® are used to make fireproof clothing. Can you guess why a blend is used rather than just one or the other? Nomex® protects firefighters, race car drivers, and monster truck and tractor drivers from getting burned should their fire-breathing rigs breathe a little too much fire. Thanks to Nomex®, an important part of American culture can be practiced safely. (Polymers play another part in the monster truck show in the form of elastomers from which those giant tires are made.)
Aramids like Kevlar® are very crystalline polymers. Because of this they are used in the form of fibers. They make even better fibers than other polyamides, like nylon 6,6. That's because of those aromatic groups we mentioned. They behave like flat connecting plates between the amide functional groups (remember those?). Because they're flat, they pack into ordered arrangements more readily, and those ordered domains are what crystals are made of. And because of the way the chains are all lined up parallel to each other in the fibers, they have incredible tensile strength, which is why they can stop a bullet.
|
Check out this 3-D model of an aramid. It's not as twisty and bent as the model of nylon right below it. This is because every other pendant group in the aramids is on the opposite side of the chain. In regular nylons the order is more random, and often the groups will get on the same side of the chain and get in each other's way. This makes the chain bend and kink so it is not as straight. As you can see when you look at the two side by side, the aramids are more regular and spread out, so they interlock better to form very good strong fibers.
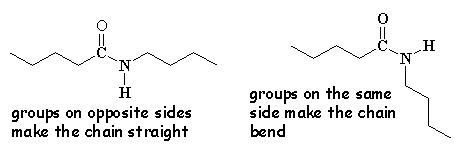
Aramids are so straight and crystalline that they have to exist as fibers and they won't even melt, so they have to be processed in special ways. |
| Other polymers used as plastics include: | Other polymers used as fibers include: |
| Polypropylene | Polypropylene |
| Polyethylene | Polyethylene |
| Polystyrene | Nylon |
| Polycarbonate | Kevlar and Nomex |
| PVC | Polyacrylonitrile |
| Nylon | Cellulose |
| Poly(methyl methacrylate) | Polyurethanes |

|
Return to Kinds of Polymers |

|
Return to Main Page |