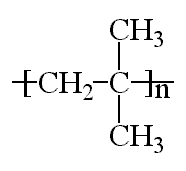
The model on the above is an image of the mol model you can
view by clicking here or you can just click on the image itself.
Either way, be sure to close the new window that opens up
with the 3D model in it when you are ready to come back here.
Polyisobutylene is a synthetic rubber, or elastomer. It's special because it's the only rubber that's gas impermeable. That is, it's the only rubber that can hold air for long periods of time. You may have noticed that balloons will go flat after a few days. This is because they are made of polyisoprene, which is not gas impermeable. Because polyisobutylene will hold air, it is used to make things like the inner tubes, liner layers of tires, and the inner liners of basketballs.
One of the fascinating things about PIB (fascinating if you're a polymer scientist anyway) is that it should be crystalline but it's not. Most polymers with highly symmetrical backbones like PIB are crystalline. Take isotactic polypropylene or polystyrene. Both are high melting, crystalline polymers. And if not crystalline, what about Tg? If we compare other polymers with and without a disubstituted carbon in the backbone, the ones with have much higher Tg's, way above room temperature. Take poly(methyl acrylate) and poly(methyl methacrylate), for example. The latter has a Tg much above room temperature, around 120 oC while the former is around or just below ambient. I'm not sure what the explanation is for the unusual properties of PIB is but I'm sure we'd all like to find out.
Polyisobutylene, sometimes called butyl rubber, and other times PIB, is a vinyl polymer. It's very similar to polyethylene and polypropylene in structure, except that every other carbon is substituted with two methyl groups. It is made from the monomer isobutylene, by cationic vinyl polymerization.
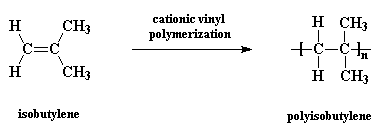
And this is that monomer isobutylene:
The model on the above is an image of the pdb model you can
view by clicking here or you can just click on the image itself.
Either way, be sure to close the new window that opens up
with the 3D model in it when you are ready to come back here.
Usually, a small amount of isoprene is added to the isobutylene. The polymerization is carried out at a right frosty -100 oC, or -148 oF for you Americans out there. This is because the reaction is so fast we can't control it unless we freeze it colder than a brass toilet seat in the Yukon.
Polyisobutylene was first developed during the early 1940s. At that time, the most widely used rubber was natural rubber, polyisoprene. Polyisoprene was an excellent elastomer, and easy to isolate from the sap of the hevea tree. Huge plantations thrived in Malaysia and grew hevea trees to supply the world's rubber needs. There was only one slight problem, and that was that Malaysia had just been conquered by the Imperial Japanese Army. And wouldn't you know, we just so happened to be fighting the Second World War against them right at that moment. Before the war was over, more than sixty million people would be dead. Deprived of natural rubber, the Allied nations did some quick thinking and came up with PIB. It obviously worked, because it helped the Allies win the war.
Oh, and natural rubber certainly made a comeback. There are now enormous hevea tree farms that appear to be enough to supply the worlds appetite for natural elastomers. But synthetic elatomers are produced today at roughly 20% greater amounts (see the polyisoprene page for more details).
Ok, we didn't actually invent polyisobutylene during the war. It had been invented long before the war by chemists in Germany. There's irony for you! But it wasn't very useful until American chemists came up with a way to crosslink it. What they did was to copolymerize isobutylene with a little bit, say, around one percent, isoprene. This is isoprene:
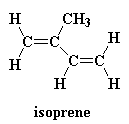
When isoprene is polymerized with the isobutylene, we get a polymer that looks like this:
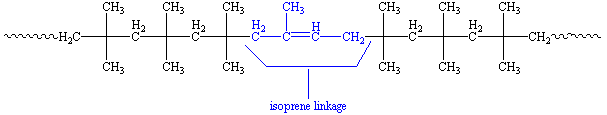
About one or two out of every hundred repeat units is an isoprene unit, shown in blue. These have double bonds, which means the polymer can be crosslinked by vulcanization just like natural rubber. What is this vulcanization you speak of? To find out, click here.
Tested Polyisobutylene Synthesis
So you want to make your own polyisobutylene. It's your lucky day! We actually have a tested method for lab scale synthesis. Be aware, though, that volatiles and flammable catalysts require extreme care in the lab to be safe!this procedure gives linear PIB: click here to see the procedure and here to download a copy.
Other polymers can be used as rubber, too. They each have their special combination of properties that makes each suitable for specific applications. Here are some of the other commercial elastomers:
- polyisoprene
- polybutadiene
- poly(styrene-butadiene-styrene)
- polychloroprene
- polyurethanes
- acrylate copolymers

|
Return to Level Two Directory |

|
Return to Macrogalleria Directory |

