
Keywords
elastomer,
gel,
thermoset
A long time ago...how long, you ask? It was about a hundred and sixty years ago, 1839 to be exact. This was before there were any cars to need tires, but the idea of a useable rubber was still attractive. One person trying to make rubber more useful was named Charles Goodyear, a tinkerer and inventor, and by no means a successful one at this point. While goofing around in his kitchen with a piece of fabric coated with a mixture of rubber latex, sulfur and a little white lead, he accidentally laid it on a hot stove top. It began sizzling like a mass of really smelly bacon or (strangely enough) burning rubber. Wouldn't you know, when he took a look at this mass of rubber, he found it wouldn't melt and get sticky when it was heated, nor would it get brittle when he left it outside overnight in the cold Massachusetts winter. He called his new rubber vulcanized rubber.

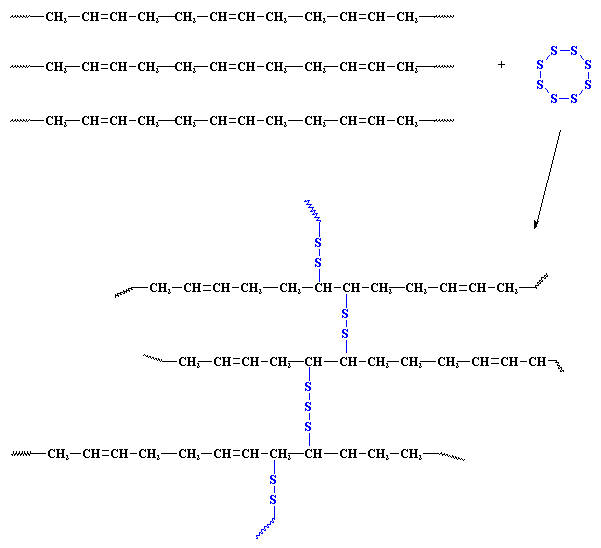
Yes, folks, this means exactly what you think it does. An object made of a crosslinked rubber is in fact one single molecule. A molecule big enough to pick up in your hand.
Wow!
These crosslinks tie all the polymer molecules together. Because they are tied together, when the rubber gets hot, they can't flow past each other, nor around each other. This is why it doesn't melt. Also, because all the polymer molecules are tied together, they aren't easily broken apart from each other. This is why the Charles Goodyear's vulcanized rubber doesn't get brittle in when it gets cold.
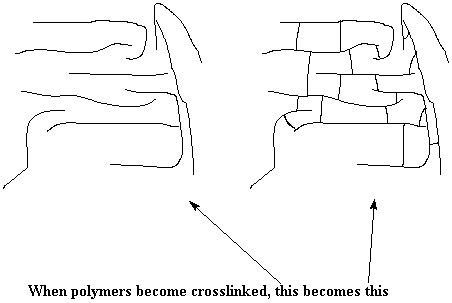
We can look at what's going on conceptually, and take a look at the bigger picture. The drawing below shows the difference between a lot of single uncrosslinked polymer chains, and a crosslinked network.
Other kinds of rubber, which chemists call elastomers that are crosslinked include:
Crosslinked polymers are usually molded and shaped before they are crosslinked. Once crosslinking has taken place, usually at high temperature, the object can no longer be shaped. Because heat usually causes the crosslinking which makes the shape permanent, we call these materials thermosets. This name distinguishes them from thermoplastics, which aren't crosslinked and can be reshaped once molded. Interestingly, the first thermoset was again polyisoprene. The more sulfur crosslinks you put into the polyisoprene, the stiffer it gets. Lightly crosslinked, it's a flexible rubber. Heavily crosslinked, it's a hard thermoset. (It was Charles' brother Noah who made the first polyisoprene thermosets, by the way.) Here are some crosslinked thermosets:
Crosslinked polymers can also be coatings, adhesives, and electronic parts. Crosslinked materials can't dissolve in solvents, because all the polymer chains are covalently tied together. But they can absorb solvents. A piece of a crosslinked material that has absorbed a lot of solvent is called a gel. One kind of gel you may be familiar with is crosslinked polyacrylamide. Uncrosslinked polyacrylamide is soluble in water, and crosslinked polyacrylamides can absorb water. Water-logged gels of crosslinked polyacrylamides are used to make soft contact lenses.
Charles Goodyear never got rich from his invention. He spent his life in lawsuits over patent infringement. But after he died, a company founded in his name, Goodyear, became the successful corporate giant we know today. The company is now centered in Akron, Ohio. I say this because across town, at another rubber company, B.F. Goodrich, someone else came up with a clever new way to crosslink rubber. Click here to find out what it was.

|
Return to Level Three Directory |

|
Return to Macrogalleria Directory |