
The model on the right above is an image of the pdb model you can view by clicking here or you can just click on the image itself.
Either way, be sure to close the new window that opens up with the 3D model in it when you are ready to come back here.
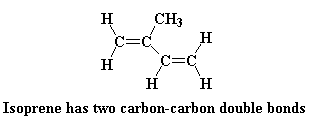
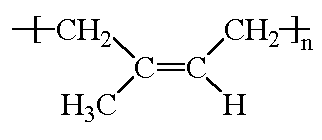
Polyisoprene is diene polymer, which is a polymer
made from a monomer containing two carbon-carbon double bonds. Like
most diene polymers, it has a carbon-carbon double bond in its backbone
chain. Polyisoprene can be harvested from the sap of the hevea tree, but
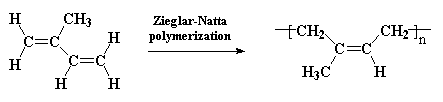
it can also be made by Ziegler-Natta
polymerization. This is a rare example of a natural polymer that we
can make almost as well as nature does.
This is what the isoprene monomer looks like:
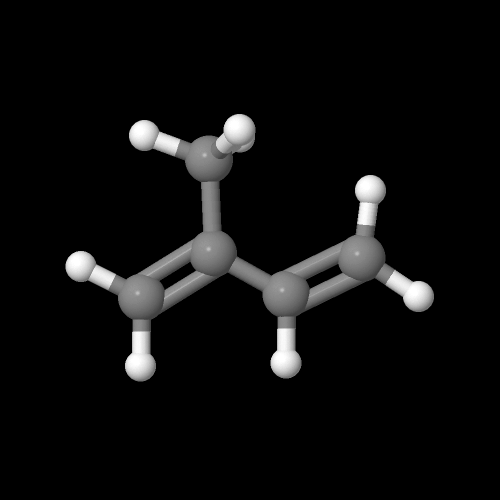
The model on the right above is an image of the pdb model you can view by clicking here or you can just click on the image itself.
Either way, be sure to close the new window that opens up with the 3D model in it when you are ready to come back here.
Other polymers used as elastomers include:

|
Return to Level Two Directory |

|
Return to Macrogalleria Directory |