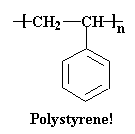

I suppose that if we're going to have a page about something called "tacticity", it might be a good idea to let you the netsurfer know just what tacticity is. Tacticity is simply the way pendant groups are arranged along the backbone chain of a polymer. We talk about tacticity a lot when dealing with vinyl polymers. To illustrate tacticity, we're going to use one of those vinyl polymers, our good friend polystyrene.
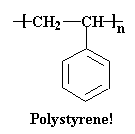
Now, polystyrene is often drawn as a flat picture like this:
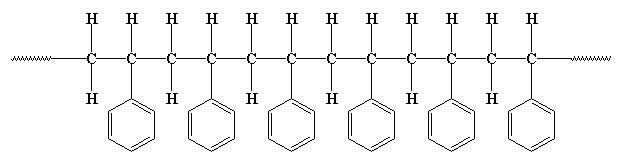
But polymers aren't really flat like that. The carbon atoms aren't really in a straight line like that, nor are the hydrogens and phenyl groups all placed at perfect right angles. The carbon chain is more of a zigzag like this:
The pendant groups tend to be point away from the chain, like this:
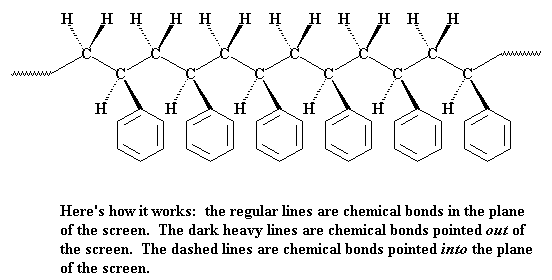
In that picture you see all the phenyl groups are located on the same side of the polymer chain. But they don't have to be this way. To illustrate let's look at a chain of polystyrene from above. You can see that the pendant phenyl groups can be either on the right or left side of the chain. If all of the phenyl groups are on the same side of the chain, we say the polymer is isotactic. If the phenyl groups come on alternating sides of the chain, the polymer is said to be syndiotactic. If the phenyl groups are on both sides and right and left follow in no particular order, in a random fashion, than we say the polymer is atactic.
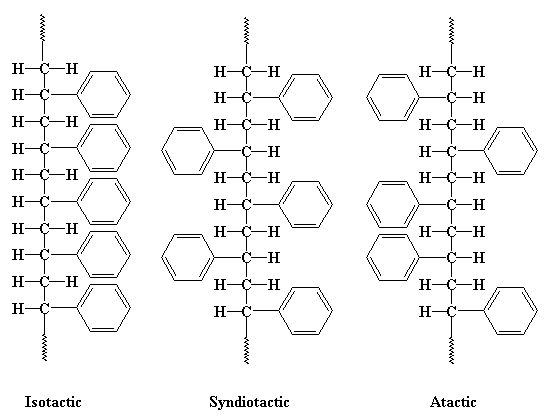
If you want to get a look at this in 3-D, click here. (Note: Isotactic polystyrene isn't produced commercially. We're just using it as a hypothetical example. Other polymers can be isotactic, though.)
What does this have to do with anything, you ask? A lot! You see, when polymers have a regular arrangement of their atoms, like we see in the isotactic and syndiotactic polystyrene, it is very easy for them to pack together into crystals and fibers. But if there is no order, as is the case with the atactic polystyrene, packing can't occur. This is because molecules pack best with other molecules of the same shape. Try packing a box full of identical objects, and different objects, and you'll get the idea.
Another vinyl polymer, polypropylene, is a good example of the effects of tacticity. At first, there was only atactic polypropylene. It's kind of soft and sticky, not very strong, and not much good for anything. Then along came two scientists named Robert L. Banks and J. Paul Hogan. They invented a new type of vinyl polymerization which ended up being named Ziegler-Natta polymerization. This new process could make isotactic polypropylene. This new polypropylene could crystallize, and could be used to make fibers for things like indoor-outdoor carpeting.

|
Return to Level Three Directory |

|
Return to Macrogalleria Directory |